2022-02-15 10:47:24
In the biochemistry and molecular biology experiments, there is a kind of buffers in alkaline range, which is higher than the commonly used physiological range, that is cyclohexylamino buffer family—2-(Cyclohexylamino)ethanesulfonic acid (CHES) and N-Cyclohexyl-3-aminopropanesulfonic acid (CAPS). Both of which are zwitterionic buffers and are not complexed with metal ions, and are also suitable for alkaline ranges. Then what is the difference between CHES and CAPS? This article will summarize it.
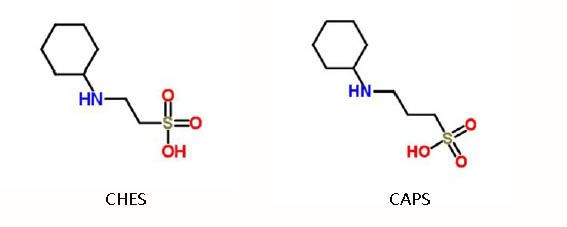
2-(Cyclohexylamino)ethanesulfonic acid (CHES)
The molecular formula of CHES is C8H17NO3S, with pKa value of 9.5 at 25°C and pH buffer range of 8.6~10.0, which is a zwitterionic buffer used in biochemistry and molecular biology. Although it is not introduced by Good et al., It is still called Good Buffer. Its applications are as follows:
(5) CHES can inhibit the viability of Micrococcus luteus at pH 9.5.
N-Cyclohexyl-3-aminopropanesulfonic acid (CAPS)
The molecular formula of CAPS is C9H19NO3S, pKa value is 10.4 at 25°C, buffer range is 9.7~11.1. The longer sulfonic acid carbon chains make it more alkaline than CHES. Although CAPS is not introduced by Good et al., it is also called as Good’s Buffer sometime. Its application areas are as follows:
(7) suitable for use with the Bicinchoninic Acid (BCA) Assay.
In summary, CHES and CAPS have a lot in common, such as the buffer ranges are alkaline, both are zwitterionic buffers, and they does not react with metal ions and so on. But there are differences between them: their buffer range is different, CAPS is more alkaline than CHES, and the applicable experiment is also different. So careful analysis is needed before use to select the most appropriate buffer.
Copyright © Suzhou Yacoo Science Co., Ltd. All Rights Reserved
Friendly Links :
online service