2017-11-28
The zwitterionic buffer, also known as Good's Buffer, is a series of N-substituted sulfamic acids that have good pH stability and are inert to a variety of chemicals and enzymes. Among them, the morpholinic buffers, including MES, MOPS and MOPSO, they do not form complexes with most metal ions and are therefore suitable for solutions containing metal ions. But what are the specific experiments they used in? This article will summarize for everyone.
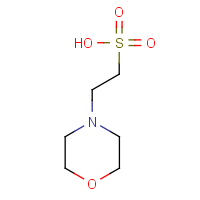
Its chemical name is 2-(N-morpholino) ethanesulfonic acid, molecular formula C6H13NO4S, pKa 6.1 at 25°C, pH buffer range 5.5~6.7. The specific available experiments are as follows:
(5) Complex with organotin (IV) molecules as antitumor agents.
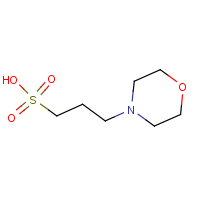
Its chemical name is 3-(N-morphine) Propanesulfonic acid, molecular formula C7H15NO4S, pKa 7.14 at 25°C, pH buffer range is 6.5~7.9. Specific available experiments are as follows:
(7) Studies on electron transport and phosphorylation of chloroplast sample preparation.
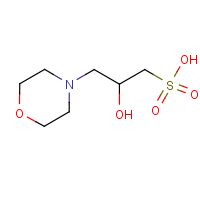
Its chemical name is 3-(N-morpholino)-2-hydroxypropanesulfonic acid, molecular formula C7H15NO5S, pKa 6.87 at 25°C, the pH buffer range 6.2~7.6. Specific available experiments are as follows:
(7) A buffer system for the determination of biological by-products of marine crude oil.
Although Good's buffer has the advantages of not participating in and not interfering with the biochemical reaction process, not inhibiting the enzymatic chemical reaction, and not reacting with the metal ion. However, in the specific use, there are still some differences in the application range of several morpholine series buffers MES, MOPS and MOPSO. Therefore, before carrying out the experiment, it is necessary to consider the buffer requirements and specific experimental types so as to select the most appropriate buffer.
Copyright © Suzhou Yacoo Science Co., Ltd. All Rights Reserved
Friendly Links :
online service